Phys. Chem. Chem. Phys., 2020,22, 14162-14168. https://doi.org/10.1039/D0CP01445A
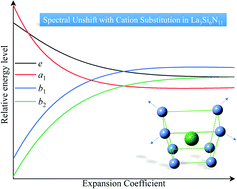
Cation substitution is a common strategy to tune the luminescence by modulating the cell parameter, polyhedral volume and bond length in solid-solution-type phosphors. Generally a close correlation between their cationic composition and spectral peak shifts can be observed. In certain compounds, however, luminescence tuning by cationic modification is almost invalid. This work is devoted to providing a reasonable explanation for the anomaly in Ce3+ doped La3Si6N11, which demonstrates unshifted excitation peaks with various cation substitutions. By simplifying the local coordination polyhedron that accommodates Ce3+ to a truncated square pyramid model, the quantitative crystal-field calculations are conducted to demonstrate the influences of the coordination environment on energy levels. The results show that the crystal-field levels become insensitive to this special type of ligand environment, leading to imperceptible peak shifts. Therefore, the relationship between the cationic composition and luminescence is determined not only by the ionic radii but also by the type of coordination polyhedron. This work shows that studying the coordination environment is helpful for achieving effective luminescence tuning.

